In 1977, Richard Peto, a British epidemiologist, theorized that large-bodied animals like elephants are more likely to contract cancer because of a higher number of cells. Yet, after researching the mortality of large mammals, scientists found his theory to be flawed. This became famously known as Peto’s Paradox.
Strangely, there was no correlation between large mammals and the risk of cancer. Researchers found it was quite the opposite. Larger, longer-living mammals actually get less cancer. In fact, elephants have a 5% cancer death rate compared to 17% for humans. There seems to be a trade off when it comes to size. But before you go bulking up, let’s delve a little deeper into the science.
So what does any of this have to do with zombies?
Well, both elephants and humans carry a sort of ‘undead’ gene called LIF6 (Leukemia Inhibitory Factor 6). It is usually dormant and is only brought back to life when a tumor is present. This is a byproduct of the gene p53, which enables the body to recognize unrepaired DNA damage.
Elephants have 20 copies of p53 whereas humans only have one, meaning elephant cells are much more sensitive to DNA damage and work faster at killing bad cells. Sometimes genes make mistakes during their duplication and produce non-functional versions known as ‘pseudogenes.’ In 2018, the pseudogene LIF6 was discovered to have an ‘on switch.’ When LIF6 is activated, it responds to damaged DNA and makes a protein that travels to a cancer cell’s mitochondria and pokes a hole in it, causing it to lose energy and eventually die.
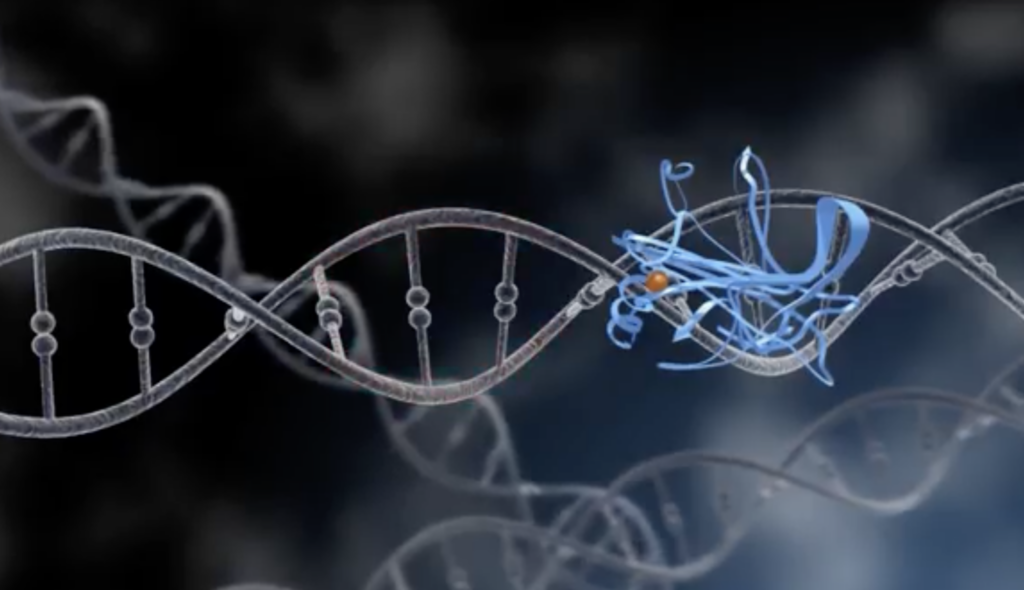
So the big, ‘elephant in the room’ question is, could we develop more zombie genes in humans to lower the risk of cancer and live biologically more like the elephant? With advancements in gene therapy, this could become a reality, but at a great cost
Gene therapy is an experimental technique that uses genes to treat or prevent disease. Doctors can treat a disorder or a disease by inserting a gene into a patient’s cells instead of using drugs or surgical treatments. There are many different approaches, like replacing mutated genes with healthy copies, deactivating a mutated gene, or introducing an entirely new gene to the body to help fight a disease.
This technology is still expensive and in its infancy. If it goes public, even people with health insurance may not be able to afford it. It’s hard to say when it will become available as there are many ethical dilemmas involved. The ultra-rich may use gene therapy to create flawless genetic babies that are super-smart athletes with good looks, giving them a huge edge over the economically disadvantaged.
For some people, this act of playing God with our bodies’ basic instructions could be seen as problematic. It could also provoke debates over who decides which genetic traits are normal and which constitute a disability or a disorder. But there are some positive outcomes.
All the billions of dollars being funneled into expensive cancer treatments could go to other important areas, like treating dementia or Alzheimer’s disease. Patients wouldn’t have to suffer through the harmful side effects of chemotherapy or cancer drugs. And we may come closer than ever to finding a permanent cure for cancer.
Although there are many potential risks and ethical implications, gene therapy would at least help us better understand how our genes work and offer a glimmer of hope. Until then, we might learn how to slow the aging process or stop the biological clock completely.